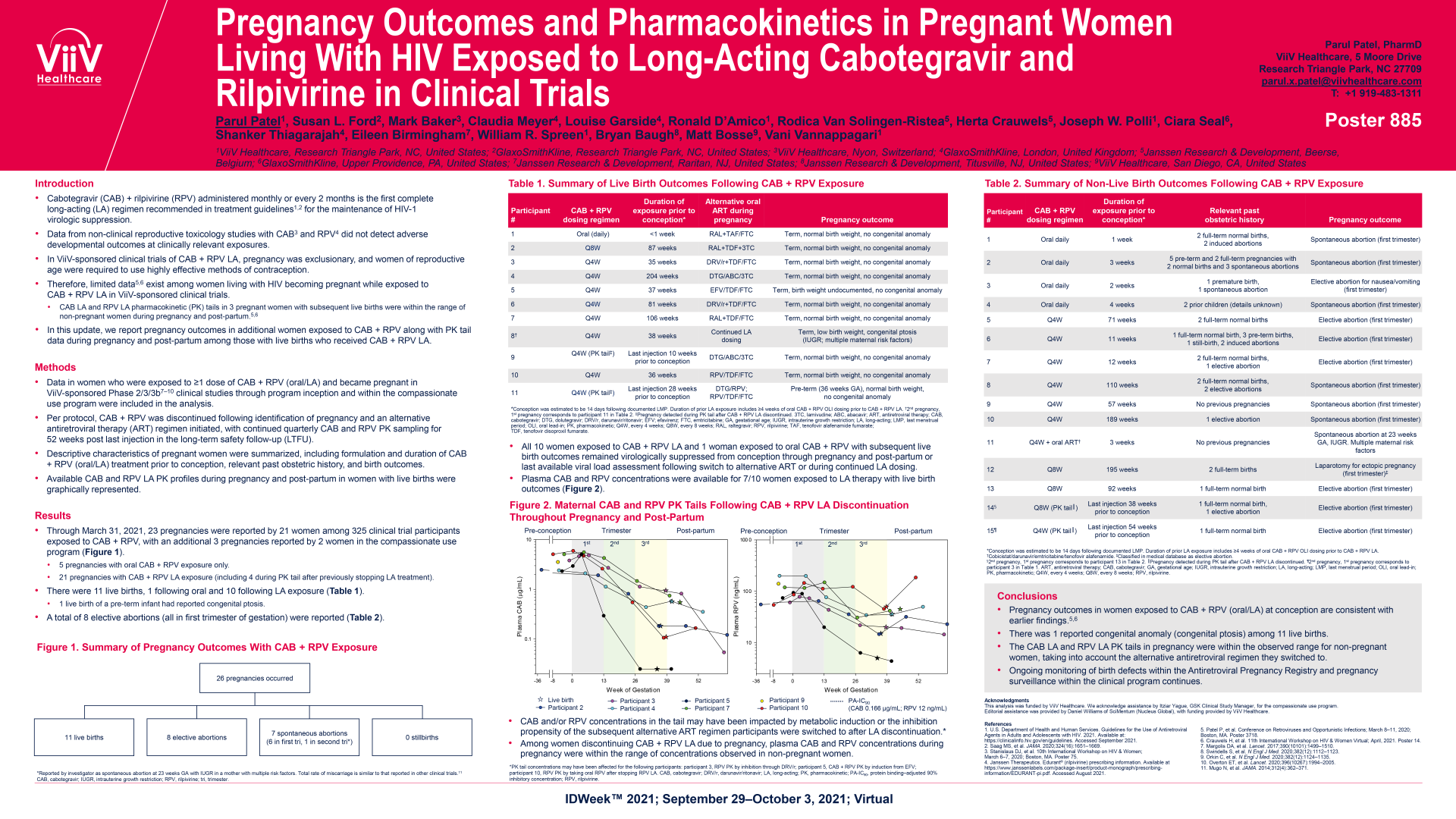
Patel P, et al. Neonatal outcomes and pharmacokinetics in pregnant women living with HIV exposed to long-acting cabotegravir and rilpivirine in clinical trials. Presented at IDWeek 2021, September 29 - October 3, 2021, Virtual Conference. Poster Presentation.
Are you a US healthcare provider?
This web portal is intended as an educational resource for healthcare providers practicing in the United States. It may include information about products or uses that have not been approved by the US Food and Drug Administration.
If you are not a healthcare provider, please discuss any questions you have regarding your health or medicines with your doctor, pharmacist, or nurse.
Connect With Our Medical Experts
Find My ViiV MSL
Easily find the ViiV Medical Science Liaison (MSL) in your area.
Talk to a Medical Expert
Connect now with a live ViiV Medical Expert.
Request a Scientific Discussion
Submit a request for additional information from a ViiV Medical Expert.
Contact Us
Chat Live
Get immediate assistance from a ViiV Healthcare Professional.
Call 1‑888‑226‑8434
Weekdays from 8 AM to 6 PM ET
(5 AM – 3 PM PT)
Report an Adverse Event
To report SUSPECTED ADVERSE REACTIONS, contact ViiV Healthcare at 1‑877‑844‑8872 or FDA at 1‑800‑332‑1088 or www.fda.gov/medwatch